xim Lifelight
Device Name:
Lifelight
Manufacturer:
xim Ltd. (Lifelight), The University of Southampton Science Park, 2 Venture Road, Chilworth, Southampton, Hampshire SO16 7NP, UNITED KINGDOM.
Measuring functions:
Blood Pressure and PR
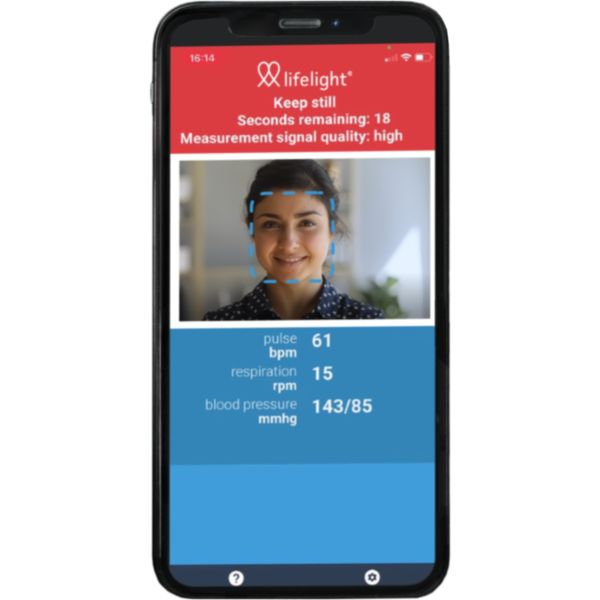
Primary Client Use:
Intended for self-measurement and home use
Measurement Site:
Face
Measurement Occurrence:
Single measurements only
Availability:
Available Currently
Description:
The xim Lifelight is a blood pressure app based on artificial intelligence. Although its blood pressure measurement technology has been evaluated, it is not possible to base a determination on its accuracy from this research. Blood pressure measurements are made, using AI, from a facial image. It is intended for self-measurement and home use.
Assessment:
The technology used in the xim Lifelight, to measure blood pressure, has been assessed, in a general population, according to a non-standard protocol.
Recommendations:
| Accuracy Assessment | Recommendation | Basis | |
| BP | Medaval | None | When an evaluation is carried out using a non-standard protocol, no inference can be made on accuracy based on a standard protocol. |
Relevant Publications:
Heiden E, Jones T, Brogaard Maczka A, Kapoor M, Chauhan M, Wiffen L, Barham H, Holland J, Saxena M, Wegerif S, Brown T, Lomax M, Massey H, Rostami S, Pearce L, Chauhan A. Measurement of Vital Signs Using Lifelight Remote Photoplethysmography: Results of the VISION-D and VISION-V Observational Studies. JMIR Form Res. 2022 Nov 14;6(11):e36340. doi: 10.2196/36340. PMID: 36374541. Available from: PMC9706384.
Ad Hoc protocol Vision-D study, (n=5152 (SBP)/ 4960 (DBP)) (Note: Error 3.5 (16.6) / -0.4 (8.5) mmHg compared to Welch Allyn Connex Spot Monitor)
Ad Hoc protocol Vision-V study, in a normobaric hypoxic chamber, (n=115 (SBP)/ 113 (DBP)) (Note: Error 2.8 (14.5) / -0.3 (7.0) mmHg compared to Welch Allyn Connex Spot Monitor)