

Distributer: Nipro Medical Europe, Blokhuisstraat 42, 2800 Mechelen, BELGIUM.
Other: Nipro Corporation, 3-9-3 Honjo-Nishi, Kita-Ku, Osaka 531-8510, JAPAN.
| Accuracy Assessment | Recommendation | Basis | |
| BG | Medaval | ★★★ Recommendation | Recent clinical validation; recent protocol; multiple clinical validations |
| BG | MDR Criteria | Self-measurement | Published evidence |
Jendrike N, Rittmeyer D, Pleus S, Baumstark A, Haug C, Freckmann G. ISO 15197:2013 Accuracy Evaluation of Two CE-Marked Systems for Self-Monitoring of Blood Glucose. J Diabetes Sci Technol. 2015 Jul;9(4):934-5. Epub: 2015 Apr 21. doi: 10.1177/1932296815582223. PMID: 25901021. Available from: PMC4525667. No abstract available.
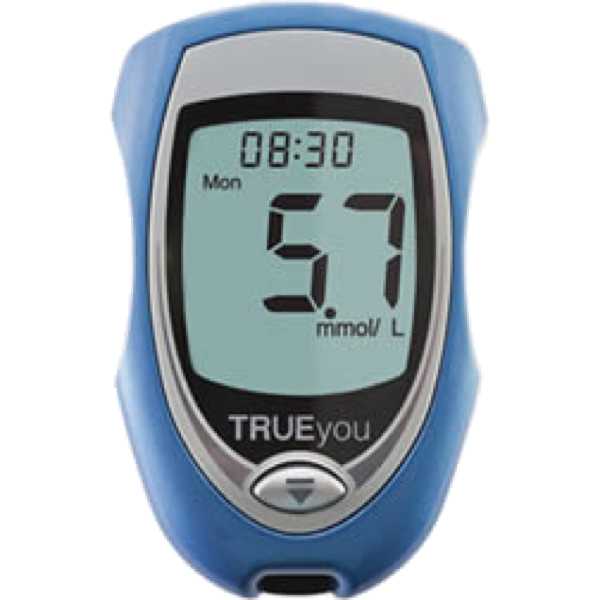
15197:2013 (aka 15197:2015) - Pass General population
Pleus S, Baumstark A, Jendrike N, Mende J, Link M, Zschornack E, Haug C, Freckmann G. System accuracy evaluation of 18 CE-marked current-generation blood glucose monitoring systems based on EN ISO 15197:2015. BMJ Open Diabetes Res Care. 2020 Jan;8(1):e001067. doi: 10.1136/bmjdrc-2019-001067. PMID: 31958308. Available from: drc.bmj.com.
15197:2013 (aka 15197:2015) - Pass General population (Note: Modified protocol: One reagent system lot.)
Freckmann G, Baumstark A, Jendrike N, Mende J, Schauer S, Link M, Pleus S, Haug C. Impact of Two Different Reference Measurement Procedures on Apparent System Accuracy of 18 CE-Marked Current-Generation Blood Glucose Monitoring Systems. J Diabetes Sci Technol. 2022 Sep;16(5):1076-1088. Epub: 2020 Aug 19. doi: 10.1177/1932296820948873. PMID: 32814455. Available from: PMC9445332.
15197:2013 (aka 15197:2015) - Pass General population
15197:2013 (aka 15197:2015) - Fail General population